Welcome to the world of yumpah which means “big medicine” in the Ute language of native Americans . We offer a wide array of strategic and tactical support to optimize clinical development timeline for the cell and gene therapy enthusiasts
Services at a glance:
Offering strategic and tactical services that include knowledge management, due diligence, audit and inspection, regulatory CMC, non clinical and clinical advice, indication prioritization, product and process development, manufacturing advice, and optimization of Target Product Profile, Provides assistance with FDA submissions including expedited designation (Fast Track, RMAT, BTD), CATT, INTERACT, PreIND, PreBLA along with Type A, C and D meetings. Provides assistance with facility design requirements, planning for surrogate end point qualification and comparability study protocols. We can also help you develop a regulatory and clinical strategy best matched to your technology to accelerate your program entry to first in human clinical trial.
-
Develop regulatory strategy matched to your technology to accelerate FIH clinical trial, perform indication prioritization and develop target product profile (TPP), align clinical and regulatory milestones with funding requirements and investor expectation. Conduct strategic due diligence and gap analysis. Provide strategic advice supporting clinical protocol development and approval based on surrogate end points.
-
Assist with FDA regulatory submissions (DMF, IDE, PMA, IND and BLA), requests for expedited designations (Fast Track, BTD, RMAT, Manufacturing Platform & Orphan Drug Designations), Assist with meetings with FDA (CATT, INTERACT, PreIND, PreBLA, Type A, C and D meetings). Assist with product, process development, manufacturing platform and analytics. Address clinical hold comments and help design comparability protocols.
-
Conduct mock prelicense inspection and perform vendor audits. Assist with facility requirements and design and Type C meeting with FDA. Assist with post approval submission supporting manufacturing and facility changes. Application of Current Good Manufacturing Practices (CGMPs) in compliance with 21 CFR 200’s, 21 CFR 600’s and 21 CFR 1271.
-
Identify best in class contract manufacturers of cell and gene therapy products. Define phase appropriate manufacturing platform and strategy. Assist with development of in house assays for characterization and release of critical starting materials, drug substance intermediates, drug substance and drug product. Assist with assay development, qualification and validation. Identify and source vendors offering compendial and or phase appropriate platform specific assays. Define suitable in process analytics technologies. Assist with development of potency assays.
-
We provide strategic advice in the following areas:
1) Regulation of HCTP derived products as a tissue product complying with 21 CFR 1271 subpart C and D (donor screening and testing and current good tissue practices CGTPs).
2) Regulation of HCTP derived product regulated as a biological drug product complying with 351 regulations.
3) Transition from 361 category to 351 products.
4) Request for Designation, Tissue Reference Group
5) Advice about combination products
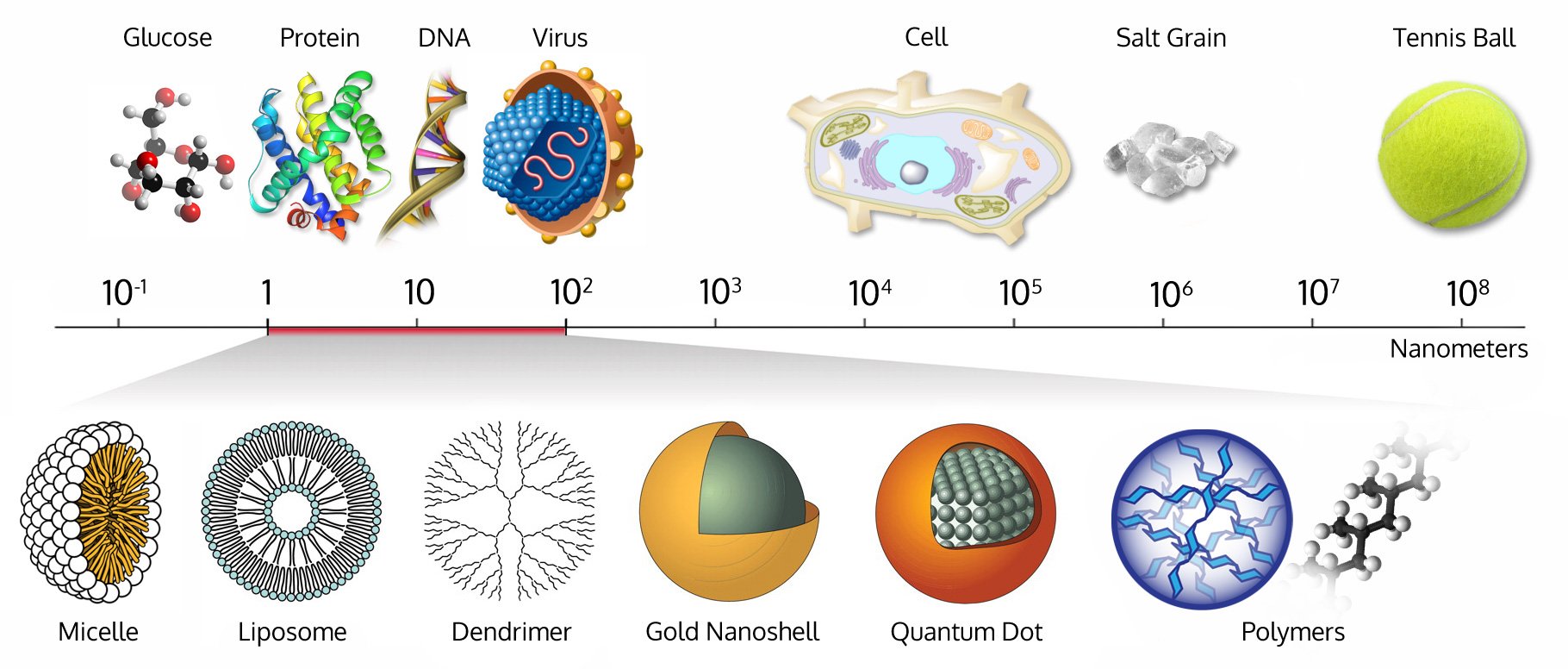
Story behind the name yumpah: When I was a kid I admired the life and integrity of native Indians watching western TV and movies. When I started working in this new space of regenerative medicine, and cell and gene therapies nearly 30 years ago I came across an Indian word that I thought it could authentically capture the spirit of discovering and developing new medicines in this field. Undoubtedly these novel classes of products are exponentially much more complicated and literally much larger than traditional small molecules. That magic word was “Yumpah or Yampah” which means “big medicine” in the Ute language (https://www.colorado.com/news/colorado-honors-its-native-peoples-events-and-offerings-educate-and-inspire). This name symbolizes and represents our mission to empower your path towards discovery and approval of innovative advanced therapies using the latest technologies and knowledge of the best practices which are developed but constantly changing in the face of transformational innovations happening in the industry and regulated by governmental agencies like FDA.
Image: Courtesy of https://www.wichlab.com/nanometer-scale-comparison-nanoparticle-size-comparison-nanotechnology-chart-ruler-2/.

